Hard water is created by groundwater passing through layers of soil, limestone, etc. thereby dissolving the ions of Ca2+, Mg2+, ... in the rock and increasing hardness in water. This is one of the parameters that need to be solved for factories using water from natural sources such as rivers, lakes ... Hard water will greatly affect the production process in textile industry by hardening and scratching fabrics as well as creating odors on products which are required high hygiene and safety, causing clogging of pipes and equipment.
Introduction of ion exchange system
To soften water, GreenWorld has developed an ion exchange system used in water treatment technology for the purpose of softening, hardness, desalination, metal removal, metal ion removal, nitrate removal, etc. through the removal of ions Ca2+, Mg2+ contained in the influent.
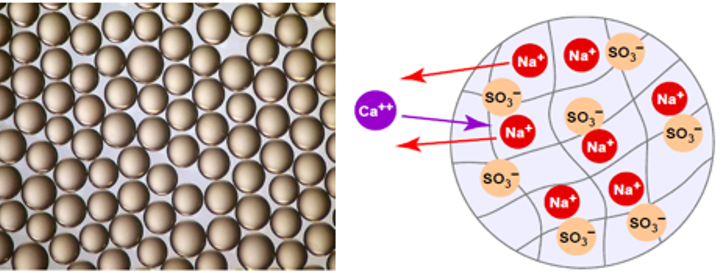
Principle of operation
In the ion exchange process, a granular substance (usually a resin) that is coated with sodium or potassium ions comes into contact with water containing calcium and magnesium ions. Two positively charged sodium or potassium ions are exchanged (released into the water) for every calcium or magnesium ion that is held by the resin. This “exchange or trade” happens because sodium or potassium are loosely held by the resin. In this way, calcium and magnesium ions responsible for hardness are removed from the water, held by the resin, and replaced by sodium or potassium ions in the water. This process makes water “softer”.
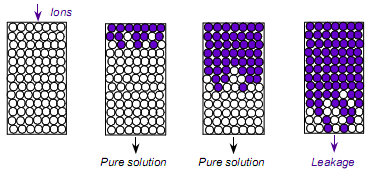
When the amount sodium or potassium ions are reduced together with plastic particles, calcium or magnesium ions in the water will not be removed. Plastics at this point are considered as being exhausted and need to be recreated by returning them to a new state and starting over. Regeneration of ion exchange resin is a reversal of the exchange reactions applied according to the characteristics of the water and the operating process.
Ion exchanger of GreenWorld
